What Are the Ethical Implications of Excessive RhIG Usage?
DOWNLOAD AND READ THE SANDLER ET AL. STUDY
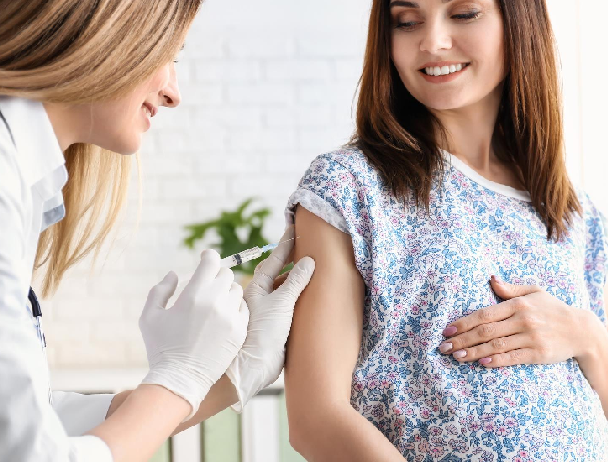
Rh immune globulin (RhIG) is a prophylactic treatment consisting of concentrated antibodies. It is given to RhD-negative women during pregnancy, and following delivery, in order to prevent them from making anti-D due to exposure to RhD-positive fetal blood. Without RhIG, the RhD-negative mother’s immune system could produce anti-D, eliciting an immune response against the RhD-positive blood of her fetus, a condition known as hemolytic disease of the fetus/newborn (HDFN).
The risk and severity of HDFN increase in subsequent pregnancies if the mother has already made anti-D. For this reason, RhIG prophylaxis is repeated for each pregnancy and delivery in RhD-negative women.
Where Does RhIG Come From?
Initially, RhIG was manufactured from stored blood collected from mothers who had experienced fetal death due to RhD alloimmunization. However, the need for RhIG quickly exceeded the available supply, and another source was needed. This demand led to the recruitment of paid RhD-negative male donors who were injected with RhD-positive red blood cells (RBCs) to maintain high anti-D titers. Blood from these donors was then pooled and used to manufacture RhIG. This process for manufacturing RhIG continues today.
Serologic Weak D Phenotypes & Unnecessary Use of RhIG
Though the testing of expectant mothers to determine their RhD status is standard practice, there are times when results are equivocal. In some patients, RhD status may be reported as positive in one test, and negative in subsequent testing.
Though these discrepancies were initially attributed to testing errors, we now know that they may be due to the presence of RHD gene variants which affect the expression of the RhD antigen on the surface of RBCs. A serologic weak D phenotype is defined as RBCs that initially are nonreactive or weakly reactive (<=2+) with anti-D, but react moderately or strongly with the addition of antihuman globulin. There are many different gene variants that may cause a serologic weak D phenotype, but only genomic testing can identify the specific gene variant responsible for the weak RhD expression.
In many facilities, patients with serologic weak D phenotypes are managed conservatively as RhD-negative. However, individuals with Weak D Types 1, 2 or 3 can be safely treated as RhD-positive. In a 2015 study by Stadler et al., it was estimated that out of approximately 16,700 obstetrics patients nationwide with serologic weak D phenotypes, 13,360 would be Weak D Types 1, 2 or 3. Because RhIG may be administered multiple times during pregnancy and following delivery, an estimated 24,700 unnecessary RhIG injections may be prevented annually with proper identification of RHD gene variants through the use of RHD genotyping.
Ethical Implications of Unnecessary RhIG Usage
While the use of RhIG has greatly reduced morbidity and mortality due to HDFN, unnecessary RhIG administration has ethical implications. First, RhIG is human-derived and obtained through paid RhD-negative male donors. Intentional alloimmunization of these donors may put them at risk if they ever need a blood transfusion and RhD-negative blood is not available.
A second concern is that a human-derived product should always be conserved for individuals who really need it. The donor risk associated with manufacturing RhIG makes its unnecessary use even more ethically questionable.
Finally, there is the financial burden of manufacturing and administering RhIG to patients unnecessarily. Costs include paying donors, the expense of creating injectable RhIG from blood product donation, and the shipping, storage and administration costs incurred by both the patient and the provider.
How RHD Genotyping Prevents the Unnecessary Use of RhIG
RHD genotyping prevents unnecessary administration of RhIG by accurately classifying serologic weak D phenotypes and guiding patient management accordingly. This one-time test can identify patients with Weak D Types 1, 2 or 3 who can be safely managed as RhD-positive. These patients do not need RhIG prophylaxis during the current pregnancy or for any subsequent pregnancy. In addition, RHD genotyping correctly identifies patients with RHD variants other than Weak D Types 1, 2 or 3, for whom RhIG is necessary.
RHD genotyping addresses ethical concerns regarding the excessive use of RhIG, while also potentially providing cost savings.
For more on RHD genotyping and it’s potential to reduce excess RhIG usage, download your copy of “It’s time to phase in RHD genotyping for patients with a serologic weak D phenotype” by Sandler et al., 2015 here.
